 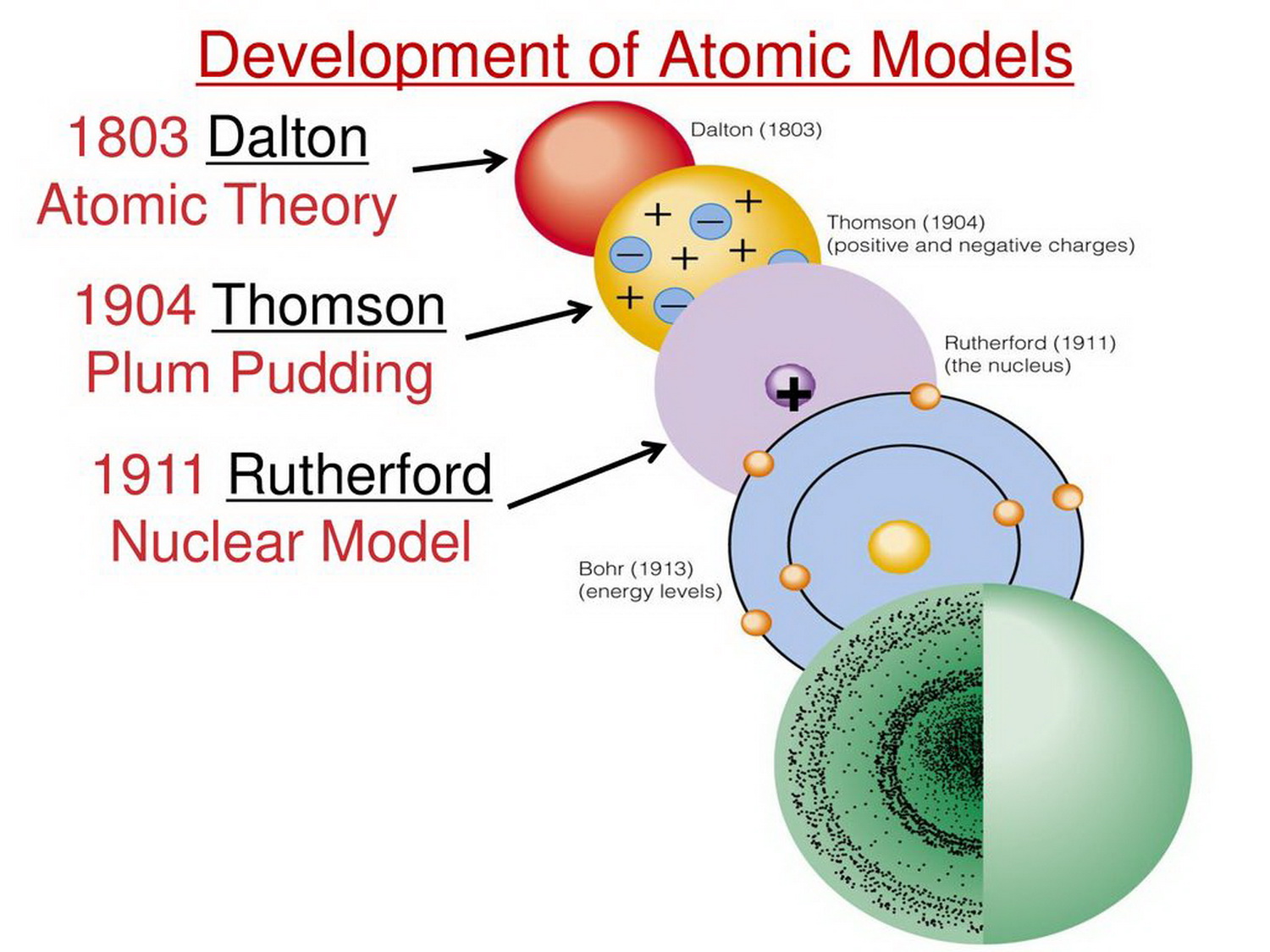  
This small volume also contains the bulk of the atomic mass of the atom. Nucleus contains relatively high central charge concentrated into very small volume. His new model introduces nucleus to the atom theory. Rutherford was first, who suggested that Thomson’s plum pudding model was incorrect. His model is often called the plum pudding model, because of his similarity to a popular English dessert. He came up with an idea that negative particles are floating within a soup of diffuse positive charge. He thought that there must be something to counterbalance the negative charge of an electron. Thomson knew that atom had an overall neutral charge. In this model, atoms were known to consist of negatively charged electrons, however the atomic nucleus had not been discovered yet. Thomson proposed his famous “plum pudding model“. Ilustration of Thomson's perception of atom Plum pudding model Īfter discovery of an electron in 1897, people realised that atoms are made up of even smaller particles. Although the awareness of atom existence goes way back to the antique period of the world history (Greek conception of atom), this article will be mainly about five basic atomic models, from which each one has somehow contributed to how we percept the structure of atom itself - Dalton´s Billiard Ball Model, J.J Thomson's "plum pudding" model, Rutherford's Planetary model, Bohr's Atomic model, Electron Cloud Model/Quantum Mechanics Model. There has been a variety of atomic models throughout history of atomic physics, that refers mainly to a period from the beginning of 19th century to the first half of 20th century, when a final model of atom which is being used nowadays (or accepted as the most accurate one) was invented. 1.6.1.2 Angular momentum quantum number: l.1.6 Basic description of the quantum mechanical atomic model:.1.5 Electron Cloud Model/Quantum Mechanics Model of Atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
